?Where is Electroplating Headed
Which way is electroplating heading?
Some recent developments, advancements, and trends in electroplating processes.
Part 1
Authors: Reza Mehtar Gharedaghi and Seyed Mehdi Bani Hosseini, Iran Electronic Board Company
In this article, we aim to provide a brief overview of the advancements and changes made in some of the important and commonly used electroplating processes that prominent companies and process developers worldwide have focused on. In summary, these changes are primarily based on the following objectives:
1. Reduction in material and metal consumption.
1- Reduction of environmental issues and elimination of hazardous pollutants from electroplating baths and resulting coatings
2- Decrease in energy consumption (cooling, heating, electricity, etc.)
3- Increase in plating speed
4- Optimization of coating properties including corrosion resistance, electrical conductivity, hardness, and wear resistance
5- Enhancement of surface brightness
6- Reduction in coating thickness
7- Ability to plate very small and intricate parts
8- Substitution of scarce and expensive materials with more economical alternatives
Preparation before coating (degreasing and rust removal):
The primary focus in these processes is on removing surfactant-containing substances such as AOX (Alkyl phenol ethoxylate) compounds that are not biodegradable and replacing them with surfactants that are biodegradable after use.
To facilitate dissolution of substances during manufacturing and prevent dust scattering in the environment, liquid degreasers are increasingly favored today, as they quickly dissolve in water, or at least introducing degreasers that require less powder for solution preparation and charging.
In the electro-degreasing processes, where gas is released on the surface of the part due to the application of electrical current, complementary cleaning operations are carried out to remove remaining contaminants from previous stages (e.g., immersion degreasing with heat). Potassium hydroxide has replaced sodium hydroxide (a caustic soda) to ensure adequate electrical conductivity of the solution even at lower concentrations and to enhance solubility.
To reduce wastewater treatment costs and maintain cleaner rinsing after degreasing, efforts are made to formulate the degreasing bath in a way that minimizes the concentration of consumables in solution preparation, while ensuring that the degreasing power remains unaffected.
Recently, new compounds have been introduced as corrosion inhibitors in acid-based solutions used for oxide removal on components. These inhibitors are introduced to prevent damage to the base metal surface, offering corrosion inhibition capabilities close to 100%. Therefore, for highly sensitive and polished components, especially those made of hardened or high-carbon steels, these solutions can be used with ease to remove only surface oxides without risking surface damage or introducing hydrogen embrittlement. This ensures that the base metals are not adversely affected by highly acidic and corrosive solutions.
Acid copper plating:
Acid copper plating is primarily used for decorative undercoats, especially in plating on plastics and electronic components, due to its high conductivity. Like other acid baths, the thickness distribution of the coating is often not uniform across the surface of the part. In some areas, the plating layer is very thick, while in others, it is very thin.
To achieve better distribution of acid copper plating uniformly across all areas of the parts, pulse plating has been replacing direct current plating and acidic copper solutions for a long time worldwide. Pulse plating, on one hand, improves the metallurgical properties of the part and, by refining the coating grain, enhances its brightness and even reduces the plating time.
This issue is prevalent in both decorative copper plating and printed circuit boards (PCBs) copper plating.
In decorative acid copper plating as well, researchers are striving to eliminate additives (brighteners) containing dyes and developing Dye Free solutions to achieve a shine similar to older solutions containing dyes while ensuring that the resulting coating is free of spots and cloudiness.
Although older brighteners containing specific dyes create shinier and smoother coatings in a shorter time, the presence of some coloring agents in their formulation darkens the plating equipment and anode bags. Therefore, it is preferred to replace them with new-generation brighteners, and the issues with these new additives are quickly being resolved.
Furthermore, the use of shield techniques has become more prevalent in plating lines than ever before. These shields are mounted on the part or jig and prevent coating deposition in high-current density areas.
In this regard, some companies have developed software that aids plating engineers in predicting the precise coating thickness distribution at each point, enabling them to achieve the best coating properties with minimal metal wastage by controlling thickness distribution throughout the part. These software tools assist engineers in optimizing the design of jigs and shields, determining the optimal distance between them and the cathode, as well as the dimensions of the plating tank, the type of agitation, and the density of the applied current. This helps them to have an accurate prediction of how parts will be plated in each area, enabling them to achieve the best coating properties with minimal metal wastage by controlling thickness distribution throughout the part.
The example below illustrates the outcome of employing this technology on brass sanitary valves. Before its implementation, there were areas with excessive coating thickness and other areas with very low thickness. This issue not only led to the wastage of copper and nickel metals during plating but also resulted in a decline in the visual appearance and corrosion resistance quality. Therefore, by utilizing electrochemical knowledge and analyzing the patterns of electrical current distribution on the part’s surface, along with changing the arrangement of parts in the tank and on jigs, using specialized jigs and shields, and adjusting the electrical current density optimally, the coating thickness distribution on the part is ideally transformed. The example below is a practical case of implementing this technique to optimize the thickness distribution for plating on sanitary valves, successfully executed by PAL Company, one of the leading manufacturers of these components.
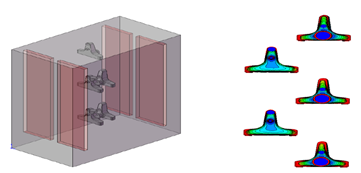
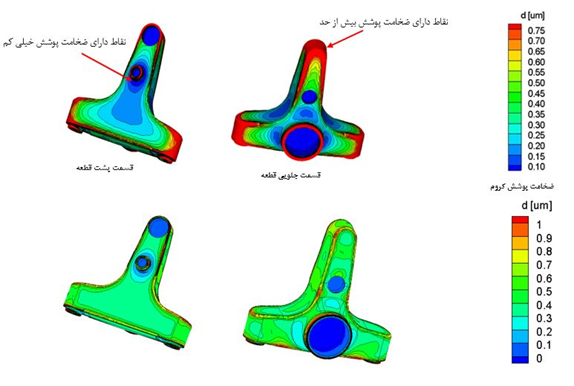
Uniform Thickness Distribution After Shielding and Proper Arrangement Against Anodes
In the realm of printed circuit board (PCB) plating, in addition to employing pulse currents, researchers are striving to develop solutions that allow for continuous horizontal plating of boards to increase production speed. In these processes, predominantly non-dissolving anodes are used, meaning copper anodes are not utilized.
Considering the reduction in the size of the boards and the drastic decrease in the existing holes, especially in HDI (High-Density Interconnect) boards, specialized copper plating solutions have been developed with the capability of quickly and uniformly filling these holes. These solutions exhibit excellent filling properties in these very small boards. Sometimes, the size of the printed circuit boards is so small (for example, in mobile devices and other electronic components) that the area to be plated with copper is barely visible or not visible to the naked eye at all.

These points, known as blind microvias, are filled by a specialized copper plating solution with a thin and uniform copper layer to facilitate subsequent plating layers and ultimately the assembly of electronic components on the assembled board. The uniformity of the copper layer and the absence of internal stress in the functioning of the final component are crucial.
Efforts are made to ensure that these solutions are usable with both pulse and direct currents, as well as in both vertical and horizontal plating lines.
Cyanide copper plating:
Cyanide copper plating is used as a primary and fine coating for plating parts made of Zamak (dry lead) and sometimes aluminum, but as its name suggests, it contains cyanide, which is highly hazardous to living organisms and the environment. Researchers have been attempting to replace them with non-cyanide solutions that, depending on the pH and composition of the solution, can provide good adhesion for the aforementioned parts. These solutions are mainly placed within the spectrum of medium alkalinity. Although non-cyanide alkaline copper solutions are currently being used commercially, they still do not match cyanide solutions in terms of coating growth rate, flexibility, and brightness. However, the necessity of eliminating cyanide is so great that non-cyanide alkaline copper solutions with similar properties to the previous solutions will soon replace them. Prior to this replacement, researchers’ efforts in plating aim to replace sodium-based cyanide copper solutions with potassium-based ones to increase cathodic efficiency and coating growth rate, as well as to make it brighter and smoother, accompanied by the use of the Interruption technique for a shinier and smoother coating.
Today, potassium cyanide (KCN) based solutions are used instead of classic sodium cyanide (NaCN) based solutions to create highly shiny and adhesive copper coatings with a coating growth rate of approximately 1 micron per minute on Zamak parts, especially when applying the Interrupted Pulse Plating technique. The brightness of the coating produced by some of these solutions is more or less similar to the coating obtained from acidic copper baths.
Other Copper Plating Processes:
Regarding other copper plating processes, recently, additives have been introduced to increase the hardness and wear resistance of copper coatings by adding them to plating solutions, which are referred to as hardeners.
For continuous copper plating applications such as reel-to-reel plating of sheets or wires, specific additives have also been introduced. These additives, used in sulfate-based baths, produce ductile and highly pure copper coatings at very high speeds without burning or roughness.
Nickel Plating:
Nickel plating is one of the most widely used and important plating processes, which has been of significant interest to researchers in this field over the past decades due to its importance and extensive applications in both decorative and technical plating. Very shiny coatings are obtained from nickel plating solutions that are less achievable by other metals.
Most research in nickel plating has focused on removing hazardous and carcinogenic organic materials from brightening formulations and efforts to create thinner but more shiny and leveling layers to simultaneously achieve greater beauty and brightness due to the high cost of this metal. It seems that an acceptable balance has now been achieved in this regard, and further research is focused on reducing brittleness and increasing corrosion resistance and durability of the coating. Some consumable additives used in plating lines pose health risks to operators; therefore, efforts are underway to replace them with safer alternatives.
In bright nickel plating, increasing the brightness level, while enhancing the leveling power and achieving greater surface brightness, may reduce the costs of polishing and buffing processes. However, it leads to a reduction in coating thickness in recessed and blind areas, resulting in severe brittleness, roughness, blackening, and wave-like patterns on the surface. Therefore, the development and testing of new brightening agents that can effectively cover areas distant from the anode while maintaining other properties of the solution and coating are currently under study and experimentation.
Nickel plating is not limited to decorative applications only. In cases where high corrosion resistance is desired, developers focus on multi-layer nickel processes with microcracks and micropores. By reducing the thickness of these layers through increasing the electrochemical potential difference between different nickel layers, developers aim to achieve maximum corrosion resistance on the coated parts.
For example, on some automotive parts where extremely high durability is required or on components used in highly corrosive conditions, multiple layers of nickel are sometimes applied, even up to 4 different layers, each with a specific thickness ratio. These layers are typically used in the following order:
1. Semi-bright nickel
2. Bright nickel
3. High sulfur nickel
4. Microcrack or microporous nickel
Exactly, each of these layers contributes to enhancing the overall corrosion resistance of the coating and provides better protection for the underlying metal surface due to their specific properties.
Their protective mechanism is based on the electrochemical potential difference between these layers and the creation of an electrochemical cell or pile to deliberately sacrifice the intermediate layers, thereby delaying the rate of deepening and the occurrence of corrosion-induced holes from the surface of the coating towards the base metal. In fact, instead of corrosion proceeding rapidly with high intensity in a pit hole vertically, it only progresses horizontally in the sacrificial nickel layer to prevent the corrosion hole from reaching the base. Deliberate creation of multiple corrosion centers throughout the coating by creating cracks or pits causes corrosion to progress not by concentrating in a smaller number of pits but in a large number of holes created by adding specific additives to the coating, thus reducing the intensity of electric current in each corrosion pit and ultimately slowing down the corrosion rate.
The presence of these intentionally created pits or cracks can lead to a decrease in the brightness of the coating. Therefore, researchers are working on solving this issue by improving the distribution of micro-pores and micro-cracks. International standards specify certain data for the number of these features per square centimeter.
Although employing these methods creates maximum durability and resistance, the consumption of nickel increases the overall cost. Therefore, extensive efforts are underway to reduce the thickness of the layers while maintaining corrosion resistance and brightness.
Improving the number and distribution of cracks or pores intentionally created in these layers during plating, or controlling the amount of sulfur in the coating, is a focus of active researchers in this industry. For example, in the semi-bright nickel layer, the sulfur concentration or percentage should be significantly reduced and ideally brought to zero. Conversely, in the high sulfur nickel layer, by using special additives, a considerable amount of sulfur should be intentionally introduced into the coating to create sacrificial properties or to establish a suitable electrochemical potential compared to its underlying layers. The image below illustrates the sequence of these layers:
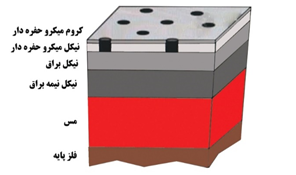
Since nickel can cause skin allergies, the use of nickel undercoating has been prohibited in industries such as eyewear, watchmaking, and jewelry like bracelets and rings that come into contact with human skin. Instead, coatings like Miralloy, a white bronze alloy, are used under the name of Nickel Free processes. Miralloy is similar to nickel in appearance but does not cause skin sensitivity in humans.
This coating consists of a lead-brass alloy or a triple alloy of lead-brass-tin, which has also become prevalent in other industries such as eyeglasses, watches, buttons, zipper making, and accessories for bags, shoes, and clothing. By changing the alloy composition, various colors such as white, gold, and gunmetal can be achieved easily.
The Miralloy process involves a cyanide-based solution for coating copper-lead (tin) in barrel plating systems. The resulting white coatings serve as suitable replacements for silver coatings in terms of tarnish resistance and corrosion resistance. Additionally, these coatings exhibit high contact resistance against sulfur-containing environments. Efforts are also underway to eliminate cyanide from this process.
The Diamagnetic properties of the coatings make them suitable for a wide range of applications. Additionally, this process is free from heavy elements, making the purification of resulting wastewater easily achievable. Some industrial applications of White Bronze coatings include:
– High-frequency connectors
– Contact elements
– Solder pins
– Battery coatings
– Cooling coils
– Hydraulic components

Chrome plating:
Chrome plating is carried out to enhance the corrosion resistance of nickel coatings and protect the surface while also creating a beautiful and unique appearance. Few coatings in the world can match the color and properties of chrome. A very thin layer of chrome, typically only 0.3 microns or less, not only provides excellent protection but also creates good abrasion resistance and shine. Its price is not significantly higher compared to other metals. However, unfortunately, chromium, especially in its hexavalent state, has significant environmental and occupational health impacts in the plating industry.
The previous plating solutions contained chromic acid or hexavalent chromium.
Nowadays, hexavalent chromium-free solutions have been developed and have effectively replaced them in decorative applications.
Although the color of the resulting coating has become almost similar to hexavalent chromium after over a decade of research and development, its corrosion resistance, especially in chloride-containing environments, is not as good as the previous solutions. Research is still ongoing to increase the corrosion resistance of coatings obtained from trivalent chromium baths.
Given the high toxicity of this metal, it is predicted that in the coming years, a larger volume of these new solutions will replace hexavalent chromium-containing solutions worldwide. Especially in Europe, many companies, particularly in Germany, have made this transition.
It’s interesting that since 1932, this issue has been of interest to researchers, so much so that the first patent for trivalent chromium plating was filed by Schlotter. Since then, significant advancements have been made in these solutions to address their shortcomings, including crack distribution, hardness, and most importantly, coating color.
It’s worth mentioning that this shift has also been attempted in hard chrome and black chrome plating solutions, but with less success. However, with the global determination to eliminate processes containing toxic hexavalent chromium compounds, it seems that even hard chrome solutions may undergo this transition in the near future. Some companies have even introduced trivalent chrome plating with a black finish to the market. For example, the plastic parts of Mercedes-Benz side mirrors are now using this coating.
Substituting chrome coatings with other metals and alloys has also been a focus, especially in East Asia, over the past two decades. Cobalt-nickel alloy, particularly in the form of a tin-cobalt alloy, has successfully replaced chrome in decorative aspects, especially in plating small Harley-Davidson parts. However, since cobalt itself is highly toxic and considered a prohibited substance, research is underway on other alloys, including tin-nickel, to make their resulting coating color more similar to chrome. In this regard, other coating processes such as PVD and plasma spraying technology, HVOF (High Velocity Oxygen Fuel) coating, tungsten carbide coating alone or with other composite particles, and some electroless nickel-phosphorus materials, are in intense competition to replace hard chrome coatings.

Part2
In the silver plating process, the majority of research and changes have been focused more on improving anti-tarnish processes (preventing the coating from darkening) to increase corrosion resistance against color change and atmospheric dulling, especially in decorative applications, rather than on the silver plating itself.
At the same time, efforts have also been made to remove antimony from silver brighteners. The formation of Sb5+ in the solution reduces the adhesion of the silver coating. Therefore, new brighteners should be as free as possible from this compound, while also introducing less sulfur into the coating structure.
Plating precious metals (expensive)
Due to a sharp increase in the prices of precious metals in the global market and, on the other hand, severe shortages of some of them such as rhodium, extensive efforts have recently been made to replace it. This metal is widely used in jewelry plating and electronic components. Over the past year, a very sudden and unexpected price hike has led research teams to actively and rapidly seek solutions for its replacement, with good successes achieved through alloying it with metals such as platinum and ruthenium, resulting in at least a 30% reduction in plating costs. Nowadays, platinum-ruthenium alloy plating (75-25) is widely used.
With colors similar to rhodium, some gold-jewelry companies have substituted it with alloys that are up to 90% cheaper than pure rhodium. These alloys must have a white color similar to rhodium and possess properties such as scratch resistance and tarnish resistance. It seems that until the price of rhodium, which is one of the most expensive precious metals, returns to previous levels, the focus will continue to be on improving the properties of these alternative alloys.
For example, in the case of black rhodium plating, an alloy of rhodium with the cheaper metal ruthenium has reduced its cost by up to 50%.
Galvanized plating:
Arguably, the most significant changes in plating processes in recent years have occurred in various aspects of galvanized plating and post-plating operations such as chromates, passivation, and various topcoats (sealers).
Apart from eliminating elements such as cyanide from galvanized plating solutions, which has been a concern for a long time and has now been effectively replaced with non-cyanide alkaline galvanizing solutions, there is also a slight amount of cyanide that unintentionally forms in some baths of alloyed zinc-nickel galvanizing (due to oxidation of some organic additives on the anode surface, leading to dullness of the coating and increased consumption of brighteners) that has caught the attention of researchers. They are focusing on using these pure resins to remove cyanide from the solution to overcome these issues.
To address environmental concerns in both acidic galvanized plating and alloyed zinc-nickel galvanizing processes, new solutions have recently been introduced that are free of boric acid and ammonia. Previously, these two substances were used as buffering agents, but now less hazardous alternatives have been introduced for environmental safety.
Following the fundamental changes in removing hexavalent chromium (Cr+6) compounds from chromate solutions, which began in the previous decade, there are currently revisions underway in substitute solutions. Even in environmentally benign passivation solutions, elements like fluoride and cobalt have been removed from the formulation so that the alternative materials can provide the same color, corrosion resistance, and adhesion standards. Additionally, the color of passivation coatings on galvanized layers can be adjusted as desired. These additives are generally referred to as color enhancers.









